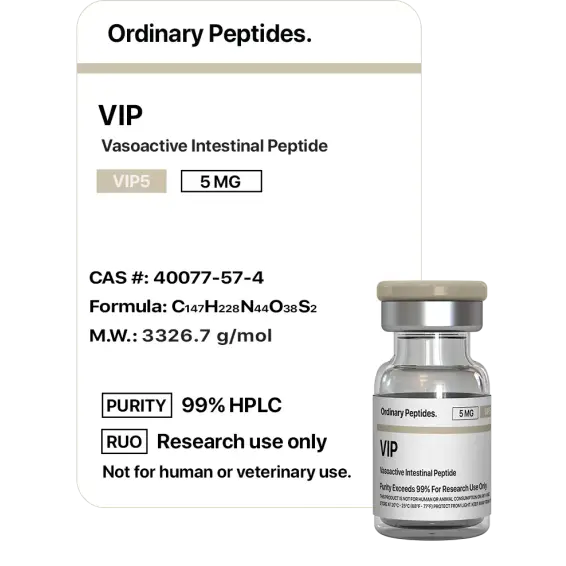
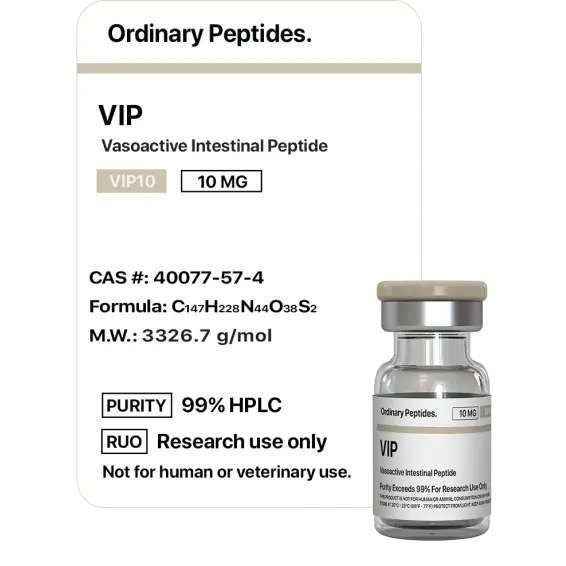
VIP
For in vitro testing and laboratory use only. Not for human or animal consumption. Bodily introduction is illegal. Handle only by licensed professionals. Not a drug, food, or cosmetic. Educational use only.
VIP (Vasoactive Intestinal Peptide): A Neuropeptide With a Broad Research Map
VIP is an endogenous 28-amino acid peptide that in a research context has long been of interest not only as a vasodilator, but as a multifunctional regulator of signals between the nervous, immune, and gastrointestinal systems. In published observations it is associated with the coordination of circadian rhythms, regulation of intestinal motility and secretion, and immune balance through effects on proinflammatory cytokines and regulatory T-cells.
In preclinical data, VIP consistently showed itself to be a potent anti-inflammatory module, which is why its therapeutic potential has been actively studied in autoimmune and pulmonary conditions. In the course of clinical investigation of the synthetic analogue aviptadil, encouraging signals were obtained in pulmonary hypertension, sarcoidosis, and respiratory failure, although this direction has not yet received full approval for routine use.
In simple terms, VIP is not a "peptide for all occasions," but a serious subject of science with a very broad physiological role. This is precisely why it is of particular interest to those who look at peptides not through the noise, but through research depth.
VIP (Vasoactive Intestinal Peptide / Aviptadil): A Scientific Review
Based on peer-reviewed literature — see References. Last updated: April 2026.
The Short Version
Vasoactive intestinal peptide is one of the most widely distributed and physiologically important neuropeptides in the human body, with roles spanning the circadian rhythm master clock, gut motility control, pulmonary vasodilation, immunomodulation of innate and adaptive immunity, pancreatic insulin secretion, and neuroprotection. Its biological reach is extraordinary.
The name is an artefact of its discovery: VIP was named for the most obvious effect observed when injected into experimental animals in 1970 — a rapid and potent reduction in blood pressure from vasodilation. The vasodilation is real and clinically significant, but it is only one dimension of a peptide that has since been identified as a master regulator at the intersection of the nervous, endocrine, and immune systems.
The pharmaceutical story centres on Aviptadil (NeuroRx, RLF-100). Aviptadil received FDA Orphan Drug Designation for pulmonary arterial hypertension (2005) and completed a Phase 2b/3 RCT in COVID-19 respiratory failure (Critical Care Medicine, 2022, N=196) that found significant oxygenation improvement and a survival signal despite missing its primary composite endpoint. A subsequent larger trial (TESICO, N=461) also failed its primary endpoint, effectively ending the COVID-19 development pathway. The central pharmacological problem preventing broader clinical development is a very short plasma half-life (~2 minutes) and systemic hypotension at doses required for non-pulmonary effects.[7]
| At a glance | |
|---|---|
| Full name | Vasoactive intestinal peptide (VIP); INN: aviptadil (synthetic form) |
| Type | 28-amino acid neuropeptide/neuromodulator/immune regulator |
| Gene | VIP gene; chromosome 6q25 in humans |
| Human sequence | Highly conserved: identical across human, cow, pig, goat, dog, rat |
| Discovery | Sami Said and Viktor Mutt; porcine duodenum; 1970 |
| Receptors | VPAC1 (broad expression), VPAC2 (CNS, smooth muscle, immune cells), PAC1 (low affinity) |
| Half-life | ~2 minutes (free peptide) — defining pharmacological barrier |
| Pharmaceutical form | Aviptadil (RLF-100, NeuroRx) — synthetic VIP |
| FDA status | Not approved; Orphan Drug Designation for PAH and ARDS |
Discovery: Said, Mutt, and the Pig Intestine
Sami Said described, for the first time in 1969, the existence of a peptide vasoactive agent with systemic vasodilator capacity present in the lungs of mammals. In collaboration with Viktor Mutt, he purified this peptide from pig lungs but only partially. Challenges in isolating it from the lungs led them to examine the intestine, since both tissues have a common embryonic origin; using porcine duodenal tissue, they isolated and sequenced the vasodilator peptide, calling it the vasoactive intestinal peptide in their 1970 Science publication.[1] Within a few years, VIP was found throughout the central and peripheral nervous systems — in neuronal cell bodies, axons, dendrites, and presynaptic endings. The “intestinal” peptide was a neuropeptide; the “vasoactive” peptide was, among many other things, a neuromodulator, immune regulator, and circadian rhythm organiser.
The VIP Peptide Family
VIP belongs to a family of homologous bioactive peptides that includes PHM, PACAP (pituitary adenylate cyclase-activating polypeptide; 27 or 38 AA), secretin, glucagon, GIP, GHRF, and helodermin. The most closely related is PACAP, sharing 68% amino acid identity with VIP and binding all three VIP receptors (VPAC1, VPAC2, PAC1) with high affinity. The structural family also includes GIP — one of the two targets of tirzepatide, covered elsewhere in this series — illustrating the evolutionary conservation and pharmacological importance of this peptide family.[2]
Structure
Sequence (human): His-Ser-Asp-Ala-Val-Phe-Thr-Asp-Asn-Tyr-Thr-Arg-Leu-Arg-Lys-Gln-Met-Ala-Val-Lys-Lys-Tyr-Leu-Asn-Ser-Ile-Leu-Asn-amide (28 residues; C-terminal amidation required for full receptor binding activity). MW ~3,326 Da; N-terminal histidine critical for receptor binding; amphipathic α-helix from approximately residue 6–28; no disulfide bonds. Identical amino acid sequence across humans, cows, pigs, goats, dogs, and rats — one of the most evolutionarily conserved neuropeptides known, indicating intense functional constraint on every residue.
Receptors and Signalling
VIP acts through three G protein-coupled receptors. VPAC1 (VIPR1): broad expression in gut, lung, liver, T lymphocytes, brain, thymus; couples primarily to Gs → adenylyl cyclase → cAMP elevation → PKA activation; the predominant receptor for most peripheral VIP effects. VPAC2 (VIPR2): more restricted to CNS (especially suprachiasmatic nucleus), smooth muscle, T cells, heart, kidney; the predominant receptor in the circadian clock; also the target for VPAC2-selective agonists in T2DM development. PAC1 (ADCYAP1R1): primarily activated by PACAP; VIP binds with ~1,000-fold lower affinity and is not the primary ligand. VIP signals with high affinity (Kd = 1 nM) through VPAC1 and VPAC2, and with low affinity (Kd = 1 μM) through PAC1.[2]
The final common pathway for VPAC1 and VPAC2 is cAMP elevation → PKA activation. cAMP is a ubiquitous second messenger; the nature of the cellular effect — vasodilation, immune suppression, insulin secretion, circadian resetting — is determined not by VIP itself but by which cells express VPAC receptors and what those cells’ downstream effector machinery looks like.
The Five Biological Roles of VIP
1. Circadian rhythm master clock
In the hypothalamic suprachiasmatic nucleus (SCN) — the mammalian master pacemaker for circadian rhythms — VIP-producing neurons are essential for synchronising the ~20,000 SCN neurons that collectively generate the ~24-hour oscillation governing sleep-wake cycles, body temperature, cortisol rhythms, immune function timing, and metabolic cycles. VIP in the SCN serves two functions via VPAC2 receptors: synchronisation of individual molecular oscillators (CLOCK/BMAL1/PER/CRY machinery) across the SCN neuronal network; and light entrainment, mediating the phase-shifting effect of the daily light-dark cycle on the body clock. VIP-deficient mice show severely disrupted circadian rhythms — individual SCN neurons continue to oscillate but become desynchronised, like an orchestra with each musician playing out of time with the others.
2. Gastrointestinal physiology
VIP is one of the most important neuropeptides of the enteric nervous system: it relaxes intestinal smooth muscle (contributing to the peristaltic wave), stimulates water and electrolyte secretion into the intestinal lumen, mediates relaxation of gastrointestinal sphincters, and exerts anti-inflammatory effects on intestinal epithelium. Pathological relevance: VIPoma (Verner-Morrison syndrome / WDHA syndrome) — rare vasoactive intestinal peptide-secreting tumours (usually pancreatic) cause massive secretory diarrhoea (3–10 litres/day), hypokalaemia, achlorhydria, and hyperglycaemia through uncontrolled VIP overproduction. VIPoma illustrates the consequences of sustained supraphysiological VIP signalling — relevant context for any therapeutic administration strategy.
3. Pulmonary vasodilation and lung biology
VIP is concentrated in pulmonary tissue and is one of the most potent endogenous pulmonary vasodilators, relaxing pulmonary arterial smooth muscle via VPAC1/cAMP, promoting lung surfactant synthesis by type II alveolar pneumocytes, protecting alveolar epithelium against inflammatory injury, and exerting anti-inflammatory effects on alveolar macrophages. VIP deficiency in pulmonary arterial hypertension (PAH): Male mice lacking the VIP gene develop moderate PAH spontaneously; VIP levels are significantly reduced in the plasma and lung tissue of patients with idiopathic PAH. This is the strongest evidence in humans that VIP deficiency contributes to a clinical disease — the basis for the aviptadil clinical development programme.[6]
4. Immunomodulation
VIP is one of the most extensively studied immunomodulatory neuropeptides. VIP has pleiotropic effects as a neurotransmitter, immune regulator, vasodilator and secretagogue; various cell types including immune cells express VIP receptors. Documented immunological effects: pro-inflammatory cytokine suppression (TNF-α, IL-6, IL-12, IL-1β, IFN-γ); anti-inflammatory cytokine promotion (IL-10, TGF-β, IL-1Ra); regulatory T cell (Treg) induction; tolerogenic dendritic cell induction; Th1/Th17 suppression; and M2 macrophage polarisation. In numerous animal models of autoimmune disease — collagen-induced arthritis, EAE (MS model), TNBS colitis, Type 1 diabetes — exogenous VIP is therapeutic and VIP-knockout animals develop worsened inflammatory disease.[2][5]
5. Pancreatic function and metabolism
VIP stimulates glucose-dependent insulin secretion from pancreatic beta cells via VPAC2 receptors, and also promotes islet β-cell proliferation through the forkhead box M1 pathway. This incretin-like activity has driven development of VPAC2-selective agonists as potential T2DM therapies that would stimulate insulin secretion without VPAC1-mediated vasodilation — one of the most active current VIP-related drug development programmes.[4]
The Aviptadil Clinical Development Programme
Pulmonary arterial hypertension (PAH)
Phase 2 RCT in 20 patients with idiopathic PAH: inhaled aviptadil (100 μg single dose during right-heart catheterisation) caused a small but significant selective pulmonary vasodilation and improved stroke volume; approximately 30% of patients experienced pulmonary vascular resistance reduction of >20%. However, 3-month administration did not demonstrate durable benefit, and the programme ended without establishing clinical efficacy. The development rights changed hands; LungRx was not successful in convincing investigators in Europe and the United States to continue PAH development.[9]
COVID-19 respiratory failure
Rationale: VIP binds VPAC-1 receptors on alveolar type II pneumocytes; SARS-CoV-2 also targets type II pneumocytes via ACE2. VIP upregulates surfactant production, blocks alveolar cell apoptosis, suppresses cytokine production, and in vitro inhibits SARS-CoV-2 replication in lung cell lines.
TESICO trial (Lancet Respiratory Medicine, 2023; N=461): 461 patients with COVID-19 hypoxaemic respiratory failure; 12-hour infusion of aviptadil or placebo daily for 3 days. Also failed its primary endpoint. Significant blood pressure lowering by aviptadil alongside higher rates of vasopressor use was reported — consistent with VIP’s vasodilatory mechanism.[8]
The honest assessment: two large trials, two primary endpoint failures, with interesting secondary biological signals. VIP’s pulmonary biology is genuine; the therapeutic window for systemic administration in critically ill patients appears very narrow. Treating critically ill patients with a potent vasodilator that causes hypotension requiring vasopressors is therapeutically complex. The COVID-19 development pathway is concluded.
The Pharmacokinetic Problem
The 2-minute plasma half-life of free VIP is the defining translational barrier. Three mechanisms of rapid degradation: DPP-4 cleavage at the Tyr¹â°-Arg¹¹ bond (the same DPP-4 targeted by sitagliptin); neutral endopeptidase 24.11 (neprilysin) cleaving multiple internal bonds; and rapid tissue peptidase degradation by lung, liver, and kidney. VIP-based drugs have not reached clinical use partly because of poor metabolic stability and poor penetration to desired sites of action.
Strategies under development: DPP-4-resistant analogues (substitutions at positions 2, 10); retro-inverso VIP (D-amino acid versions); long-acting analogues with albumin-binding moieties (similar to semaglutide’s half-life extension strategy); NEP inhibitor co-administration to extend VIP half-life ~3-fold; and local delivery via nanoparticles bypassing systemic degradation — particularly intranasal delivery for CNS/circadian applications and pulmonary delivery for PAH.
Current and Emerging Research Directions
Autoimmune disease: The most extensive preclinical VIP literature addresses rheumatoid arthritis, multiple sclerosis (EAE model), Crohn’s disease, and Type 1 diabetes. In collagen-induced arthritis, VIP treatment reduces synovial inflammation, prevents joint destruction, and induces Treg cells — replicated across multiple independent laboratories. The translational gap: the 2-minute half-life means systemic VIP requires continuous infusion or delivery system innovation. No autoimmune disease clinical trial has been completed. VPAC2-selective agonists for T2DM: the most commercially active VIP-related drug development area; would stimulate glucose-dependent insulin secretion via pancreatic VPAC2 while avoiding VPAC1-mediated vasodilation. Several candidates are in preclinical/early clinical evaluation.[4]
Safety Profile
The dominant and mechanistically inevitable adverse effect of systemic VIP administration is hypotension from vasodilation — confirmed in both COVID-19 clinical trials, with TESICO-treated patients requiring vasopressor use. Other documented effects: facial flushing, nausea (GI smooth muscle effects), transient heart rate changes, and gastrointestinal effects at higher doses. The inhaled route in PAH studies produced mainly pulmonary effects with minimal systemic vasodilation — consistent with the rationale for pulmonary-targeted delivery; adverse events were mild (cough, transient flushing). The VIPoma context establishes what sustained VIP excess produces physiologically (WDHA syndrome), providing the outer bounds of chronic VIP overactivation.
Common Misconceptions
“VIP deficiency causes or worsens CIRS/mould illness.”
VIP deficiency has been documented in pulmonary arterial hypertension (Shawn Said’s work, confirmed in animal models and human measurements). The broader claim — that VIP deficiency is a central mechanism in Chronic Inflammatory Response Syndrome attributed to biotoxin/mould exposure, and that intranasal VIP corrects it — reflects the work of a single researcher (Ritchie Shoemaker) and has not been independently validated in peer-reviewed trials. This claim should not be conflated with the established pulmonary hypertension VIP deficiency data.
“Aviptadil proved VIP works for COVID.”
Aviptadil improved oxygenation significantly and showed a survival signal in the smaller 196-patient trial. The larger 461-patient TESICO trial failed. The overall clinical verdict on VIP for COVID-19 respiratory failure is negative for primary endpoints. The biological signal is real; the clinical benefit at the dose and timing studied is undemonstrated.
“VIP would work if you could just give enough of it.”
The dose-limiting adverse effect is hypotension. Increasing the dose to achieve better anti-inflammatory effects also increases vasodilation, requiring vasopressors in already critically ill patients. The therapeutic window is narrow and has been tested clinically. This is why VPAC2-selective agonists (avoiding VPAC1-mediated vasodilation) are the preferred current drug development approach.
Key Takeaways
- VIP is a fundamentally important endogenous neuropeptide with biological roles spanning circadian biology, gut physiology, pulmonary vasodilation, immunomodulation, and metabolic regulation. Its discovery name reflects the accident of how it was found, not the full scope of its biology.[1]
- â The most clinically established VIP-related finding is VIP deficiency in pulmonary arterial hypertension. This deficiency has been demonstrated in both animal models and human patients and provides the clearest mechanistic rationale for therapeutic VIP administration.[6]
- â ï¸ Aviptadil completed a Phase 2b/3 COVID-19 RCT and failed its primary endpoint; the larger TESICO trial also failed. Oxygenation and biological signals were encouraging; systemic hypotension may have counteracted benefit. The COVID-19 therapeutic development pathway is concluded.[7][8]
- â ï¸ The 2-minute plasma half-life is the defining clinical development barrier. All promising preclinical applications require delivery system innovation or chemical modification to be clinically tractable.
- VPAC2-selective agonists for T2DM represent the most commercially active current development direction — retaining VIP’s glucose-dependent insulinotropic properties via VPAC2 while eliminating VPAC1-mediated vasodilation.[4]
References
Discovery
- Said SI, Mutt V. Polypeptide with broad biological activity: isolation from small intestine. Science. 1970;169(3951):1217–1218.
Biology and Receptors
- Delgado M, Ganea D. Vasoactive intestinal peptide: a neuropeptide with pleiotropic immune functions. Amino Acids. 2013;45(1):25–39. PMC3883350
- Therapeutic potential of vasoactive intestinal peptide and its receptors in neurological disorders. PMC2967653
- Therapeutic potential of VIP in type 2 diabetes via VPAC2. Frontiers in Endocrinology. 2022.
- A clinical approach for the use of VIP axis in inflammatory and autoimmune diseases. International Journal of Molecular Sciences. 2020.
Clinical Evidence
- Petkov V, Mosgoeller W, Ziesche R, et al. Vasoactive intestinal peptide as a new drug for treatment of primary pulmonary hypertension. Journal of Clinical Investigation. 2003;111(9):1339–1346.
- Youssef JG, Lavin P, Schoenfeld DA, et al. The use of IV vasoactive intestinal peptide (aviptadil) in patients with critical COVID-19 respiratory failure: results of a 60-day randomised controlled trial. Critical Care Medicine. 2022;50(11):1545–1554. PMC9555831
- Brown SM, et al. TESICO trial: aviptadil for COVID-19 hypoxaemic respiratory failure. Lancet Respiratory Medicine commentary. 2023.
- Inhalation of vasoactive intestinal peptide in pulmonary hypertension. PMID 18978135. PAH Phase 2 study.
Key Investigators
- Sami I. Said, MD — SUNY Stony Brook; discoverer of VIP (1970) with Viktor Mutt; pioneered the pulmonary VIP biology underpinning the PAH deficiency hypothesis; first to demonstrate VIP’s protective effects against acute lung injury.
- Mario Delgado, PhD and Elena González-Rey, PhD — IPBLN-CSIC, Granada, Spain; produced the foundational research on VIP as an immunomodulator in autoimmune models; characterised VIP’s effects on regulatory T cells, tolerogenic dendritic cells, and autoimmune arthritis/EAE.
- Jonathan Javitt, MD, MPH and Jonathan Morganroth, MD — NeuroRx; principal investigators on the COVID-19 aviptadil clinical development programme.
Vasoactive Intestinal Peptide (VIP) is a naturally occurring 28-amino acid neuropeptide and hormone first isolated from porcine duodenum in 1970. It belongs to the secretin/glucagon superfamily and is produced throughout the body — in the gut, pancreas, lungs, heart, brain, and immune system. It functions simultaneously as a neurotransmitter, neuromodulator, and neuroendocrine signaling molecule, making it one of the most broadly active peptides in human physiology.
VIP binds to two main receptors — VPAC1, found primarily in the lungs, gut, and central nervous system, and VPAC2, found mainly in the immune system and endocrine glands. Binding activates adenylyl cyclase, which increases cAMP and triggers downstream signaling through PKA and CREB pathways. This leads to vasodilation, smooth muscle relaxation, immune modulation, circadian rhythm regulation, and anti-inflammatory effects. On a molar basis it is 50 to 100 times more potent as a vasodilator than acetylcholine.
Research spans a wide range of conditions. In pulmonary medicine, VIP has shown significant results in pulmonary arterial hypertension, reducing pulmonary artery pressure and improving exercise capacity via inhalation. In gastroenterology it regulates gut motility, stomach acid secretion, intestinal wall integrity, and beneficial gut bacteria. In immunology it is being studied as a treatment for autoimmune diseases such as rheumatoid arthritis, multiple sclerosis, and inflammatory bowel disease due to its ability to suppress pro-inflammatory cytokines. It is also being investigated for Chronic Inflammatory Response Syndrome (CIRS) related to mold toxicity exposure.
Yes. VIP is a key regulator of circadian rhythms through its action on the suprachiasmatic nucleus — the brain's master clock. It synchronizes the molecular clock by activating Per1 and Per2 gene expression. It also plays a role in neuroprotection, and low VIP levels have been associated with cognitive deficits, mood dysregulation, and anxiety in animal models. It is co-released with GABA and modulates neuronal synchrony.
VIP is not broadly FDA-approved as a standalone therapeutic. Its synthetic analog Aviptadil has been studied in clinical trials for pulmonary arterial hypertension and COVID-19 respiratory failure, where it doubled 60-day survival odds in some patient groups. Administration routes under investigation include inhalation, intravenous infusion, and intranasal delivery. Intranasal VIP has been used in some specialty clinics for CIRS patients to restore grey matter brain volume.
When administered via inhalation at therapeutic doses, VIP has shown a remarkably clean safety profile — clinical studies report virtually no side effects via this route. At higher systemic doses, potential concerns include significant drops in blood pressure due to its potent vasodilatory effects, increased heart rate, and flushing. Its very short natural half-life of approximately two minutes in the bloodstream limits systemic accumulation but also requires careful delivery methods to maintain effective tissue levels.
People with low blood pressure or cardiovascular instability should exercise caution given its powerful vasodilatory properties. Those with hormone-sensitive conditions should consult a physician, as VIP interacts with the hypothalamic-pituitary-adrenal axis. Because most therapeutic applications remain experimental and outside mainstream clinical approval, use should only occur under qualified medical supervision.